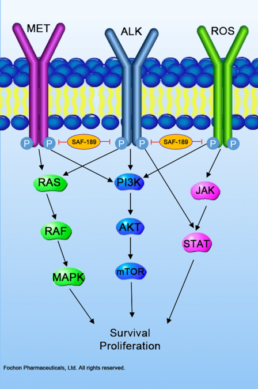
SAF-189
SAF-189 is our proprietary, oral active, highly potent and selective small molecule inhibitor of anaplastic lymphoma kinase (ALK) and ROS proto-oncogene 1 (ROS1) for the treatment of ALK-positive or ROS1-positive NSCLC. ALK and ROS1 gene rearrangements are important predictive biomarkers and oncogenic drivers for a subgroup of advanced NSCLC.
SAF-189 exhibited greatest in vitro and in vivo potency against activating and resistance mutations of ALK and ROS1 compared to approved inhibitors. It possessed superior anti-tumor efficacy, high targeted-tissue distribution to brain, lung and tumor, as well as good to excellent pharmacokinetic and safety profiles.
In the ongoing Phase 1b/2 clinical trials in China, SAF-189 exhibited good brain penetration capability and demonstrated improved efficacy and good safety profile compared to approved ALK inhibitors in crizotinib-resistant patients (including brain metastasis patients) . We believe, based on our preclinical and clinical data, that SAF-189 has the potential to be the best-in-class ALK/ROS1 inhibitor targeting ALK-positive or ROS1-positive metastatic NSCLC.
Fochon plans to leverage the clinical proof-of-concept data of SAF-189 from China to identify global partners for clinical development and commercialization.